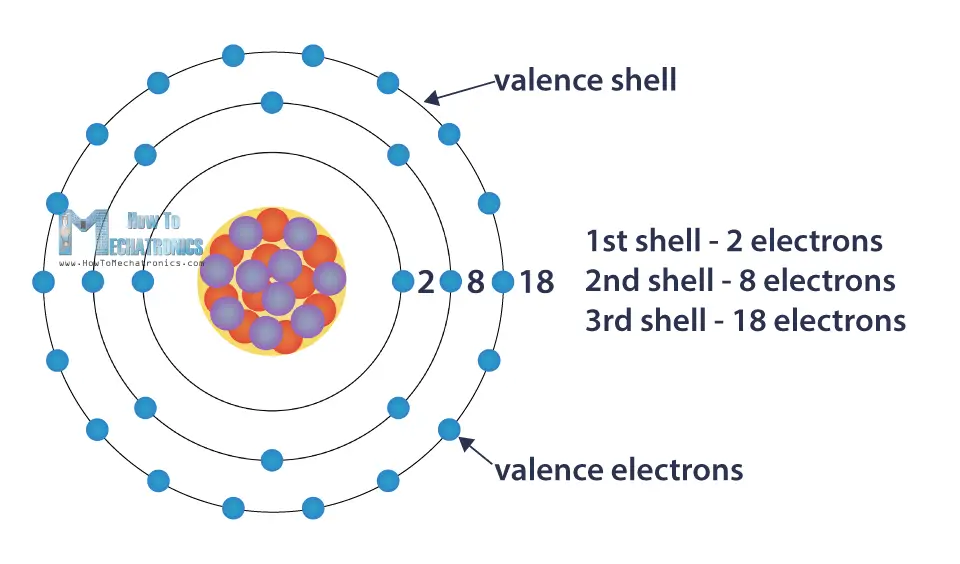
The distance between the radii is 266 p m. Beneath the molecule is the label, “B r radius equals 228 p m divided by 2 equals 114 pm.” The fourth diatomic molecule is in purple. The distance between the radii is 228 p m. As we go down the elements in a group, the number of electrons in the valence shell remains constant, but the principal quantum number increases by one each time. Beneath the molecule is the label, “C l radius equals 198 p m divided by 2 equals 99 pm.” The third diatomic molecule is in red. Electronic configuration of elements ( Data page-Wikipedia) Electronic configuration for super heavy elements ( Source) Author. As we go across a period from left to right, we add a proton to the nucleus and an electron to the valence shell with each successive element. The distance between the radii is 198 p m. The second diatomic molecule is in a darker shade of green. Beneath the molecule is the label, “F radius equals 128 p m divided by 2 equals 64 p m.” The next three models are similarly used to show the atomic radii of additional atoms. Because much of the chemistry of an element is influenced by valence electrons, we would expect that these elements would have similar chemistry-and they do. Thus beryllium has an He s2 electron configuration. Beginning with beryllium, we see that its nearest preceding noble gas is helium and that the principal quantum number of its valence shell is n 2. The distance between the centers of the two atoms is indicated above the diagram with a double headed arrow labeled, “128 p m.” The endpoints of this arrow connect to line segments that extend to the atomic radii below. They all have a similar electron configuration in their valence shells: a single s electron. Solution: The group 2 elements are in the s block of the periodic table, and as group 2 elements, they all have two valence electrons. 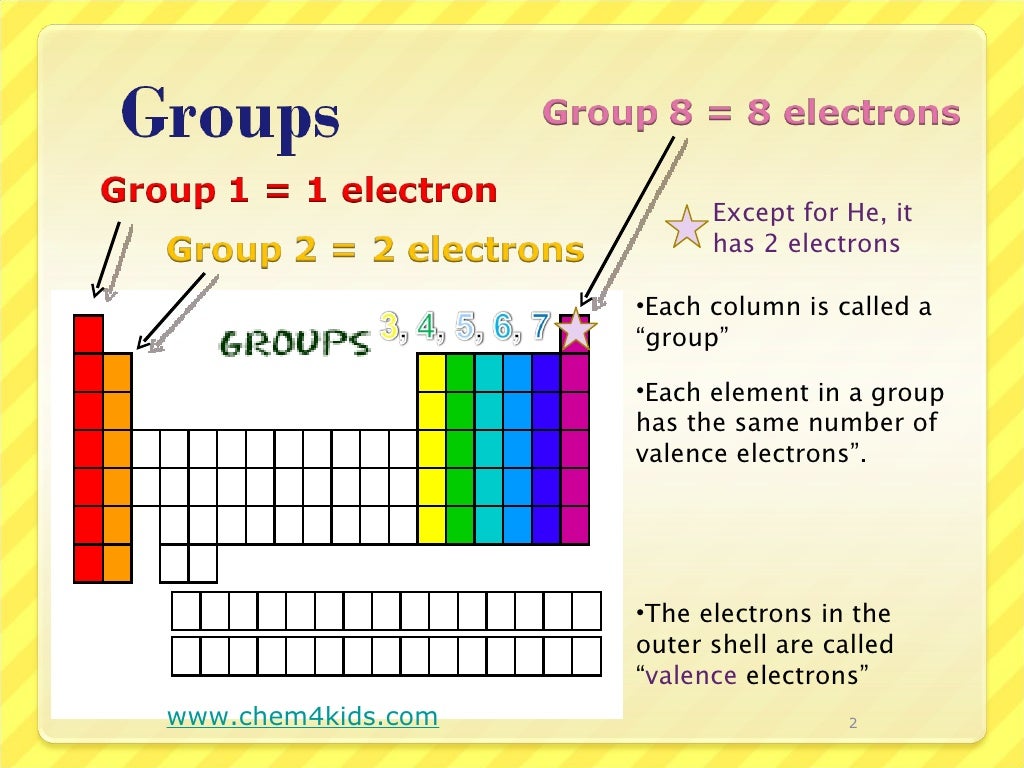
Two spheres are pushed very tightly together. The first model, in light green, is used to find the F atom radius. 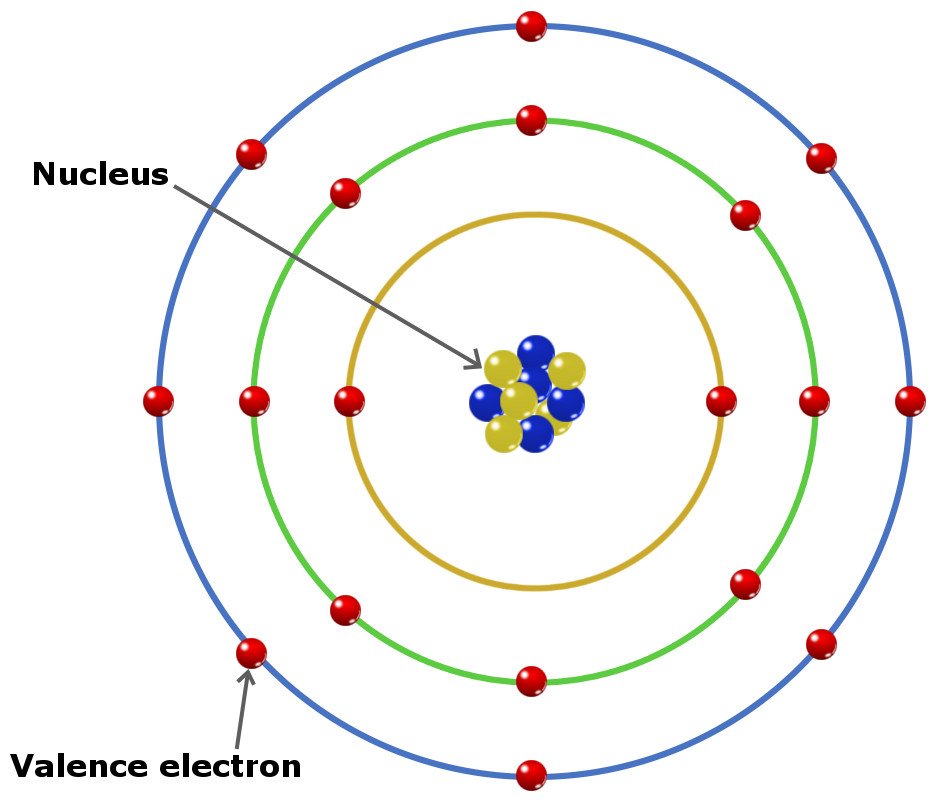
In figure a, 4 diatomic molecules are shown to illustrate the method of determining the atomic radius of an atom. Electronic configuration for lead can be written. The distribution of electrons is 2, 8, 18, 32, 18, and 4. This element has 82 number of electrons present in its 6 orbits. This metal is silvery and slightly blue but turns to be dull grey when came in contact of air. The general trend is that radii increase down a group and decrease across a period. This metal has a low melting point and soft and malleable. (b) Covalent radii of the elements are shown to scale. And so for this video, were only talking about the valence electrons for elements in the main groups. The atomic radius for the halogens increases down the group as n increases. \): (a) The radius of an atom is defined as one-half the distance between the nuclei in a molecule consisting of two identical atoms joined by a covalent bond.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
